Processing is regarded as one of the most energy-intensive stages in a polymetallic nodule value chain. Conventional assessments typically assume pyrometallurgical routes, which benefit from existing smelting capacity but also account for a large share of emissions associated with deep-sea mineral production.
A collaborative project involving TU Bergakademie Freiberg, the German Federal Institute for Geosciences and Natural Resources (BGR), and Aker BP is exploring alternative hydrometallurgical approaches that could significantly reduce energy use and environmental impact.
Their work focuses on solid-state chlorination using crystalline ammonium chloride (NH₄Cl) to extract metals from polymetallic nodules. When heated, ammonium chloride decomposes into ammonia and hydrochloric acid, which drive the chlorination process. Upon cooling, excess gases recombine to form ammonium chloride again, allowing the reagent to be reused within a closed loop.
The laboratory-scale process enables the production of concentrates of manganese, copper, nickel, zinc and cobalt. Excess ammonia can also be captured and converted into fertiliser, while metal chlorides are transferred to leaching and electrowinning stages for metal recovery.
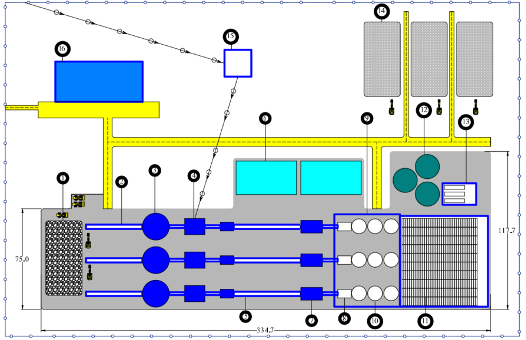
Based on these results, the researchers conducted a preliminary economic assessment of a full-scale processing facility capable of treating three million tonnes of nodules annually, matching early production scenarios proposed for commercial nodule mining operations such as those planned by The Metals Company. The proposed flowsheet (see figure above) integrates drying, mechanical processing, chlorination, leaching and electrowinning units within a centralized plant design.
A second approach explored in the study uses ammonium peroxodisulfate as an oxidising agent to improve the leaching efficiency of manganese crusts and seafloor massive sulphides found in the Norwegian Exclusive Economic Zone.
Compared with commonly used hydrogen peroxide, this compound offers higher redox potential and greater thermal stability. Laboratory results show improved oxidation of manganese minerals and increased recovery of metals such as cobalt, molybdenum, vanadium, selenium and tungsten.
The oxidising agent can be regenerated electrochemically, allowing the process to be powered by renewable energy sources such as hydropower. Inorganic residues from the process can also be converted into geopolymers, a material considered a potential low-carbon alternative to cement.
According to the researchers, these laboratory-scale methods demonstrate the potential for climate-neutral, zero-solid-waste processing of deep-sea minerals while recovering the full metal content of nodules, crusts and massive sulphides.
The work will be presented by Professor Markus Bertau (TU Bergakademie Freiberg) at Seabed Minerals 2026 in Bergen, March 24–26.

